Sulfuric acid has many uses. It is widely used in metal manufacturing in the fabrication of copper, zinc. Additionally, it is used in cleaning steel sheet metal in a process known as pickling. After pickling of sheet metal, the sheet metal is covered with a thin layer of tin, which allows the material to be used to make cans for canned food. It is also used to create titanium dioxide, which is a pigment. The sulfuric acid is used to make caprolactam, which is converted to polyamide 6, and in turn creates the titanium dioxide. Another use of sulfuric acid is the manufacture of hydrofluoric acid and phenol with propanone, which are widely used in industrial applications.

The annual production of sulfuric acid is approximately 200 million tons. The united States accounts for around 15 percent of this production. Europe accounts for around 10 percent, while Russia accounts for a little under 5 percent.
The industrial process of creating sulfuric acid is broken down into four stages. Below is a list of the 4 stages with the corresponding chemical reaction formula:
1. The Extraction of Sulfur
 2. The Conversion of Sulfur to Sulfur Dioxide
2. The Conversion of Sulfur to Sulfur Dioxide
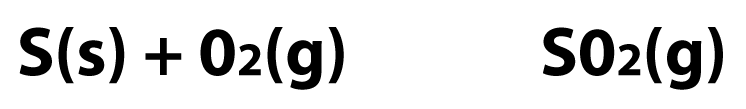 3. The Conversion of Sulfur Dioxide to Sulfur Trioxide
3. The Conversion of Sulfur Dioxide to Sulfur Trioxide
 4. The Conversion of Sulfur Trioxide Sulfuric Acid
4. The Conversion of Sulfur Trioxide Sulfuric Acid
![]()
